|
3/13/2024 0 Comments Electron group geometry chart 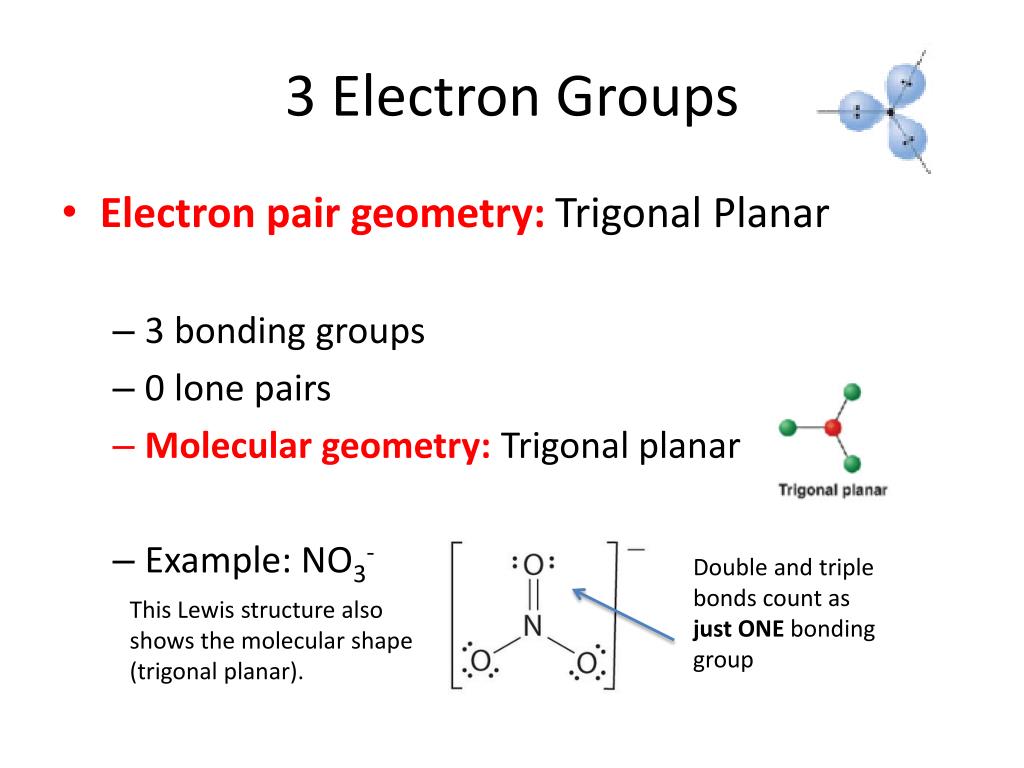
With two bonds and no lone pairs of electrons on the central atom, the bonds are as far apart as possible, and the electrostatic repulsion between these regions of high electron density is reduced to a minimum when they are on opposite sides of the central atom. The Lewis structure of BeF 2 ( Figure 7.15) shows only two electron pairs around the central beryllium atom. Other interactions, such as nuclear-nuclear repulsions and nuclear-electron attractions, are also involved in the final arrangement that atoms adopt in a particular molecular structure.Īs a simple example of VSEPR theory, let us predict the structure of a gaseous BeF 2 molecule. We should understand, however, that the theory only considers electron-pair repulsions. VSEPR theory predicts the arrangement of electron pairs around each central atom and, usually, the correct arrangement of atoms in a molecule. The electrostatic repulsion of these electrons is reduced when the various regions of high electron density assume positions as far from each other as possible. The electrons in the valence shell of a central atom form either bonding pairs of electrons, located primarily between bonded atoms, or lone pairs. The VSEPR model assumes that electron pairs in the valence shell of a central atom will adopt an arrangement that minimizes repulsions between these electron pairs by maximizing the distance between them. Valence shell electron-pair repulsion theory (VSEPR theory) enables us to predict the molecular structure, including approximate bond angles around a central atom, of a molecule from an examination of the number of bonds and lone electron pairs in its Lewis structure. H 2O is a polar molecule because the two lone pairs on oxygen mean the molecule is non symmetrical.Figure 7.14 Bond distances (lengths) and angles are shown for the formaldehyde molecule, H 2CO. Same with the C-Cl bond in CCl 4, no net dipole because the molecule is symmetrical. Both dipoles cancel and the result is no overall dipole. The C-O bond is polar because there's an electronegativity difference between C and O, but CO 2 is a non polar molecule because there is no net dipole as the molecule is symmetrical. 
You've said you know it but I'll just remind you if you already know that not all polar bonds lead to polar molecules. If you look under tetrahedral geometry, 2 bonding regions + 2 lone pairs you'll see the molecular geometry is "bent".īH 3 has 3 electron groups and 3 chemical bonds, look at the same table and the name is the same as the electron geometry, trigonal planar. You've probably seen a table of all of the names like this. SBr 2 has 4 electron groups, but only 2 of them are in chemical bonds. Molecular geometry is the geometry of the bonded atoms. Number of bonded atoms is straight forward. I'm not a mind reader though so don't yell at me if they wanted something more accurate.īH 3 has a trigonal planar electron geometry and bond angles of exactly 120°. BUT SBr 2 also has 2 lone pairs which pushes the Br-S-Br bond angle closer together, Google will tell you the exact number but <109.5° should be what they're looking for. SBr 2 has a tetrahedral electron geometry and tetrahedral bond angles are ~109.5°. In BH 3 there are 3 groups so it is trigonal planar.Ģ = linear, 5 = trigonal bipyramidal, etc.īond angles come from the electron geometry, although I don't think they want you to actually calculate it just estimate it based on the geometry. 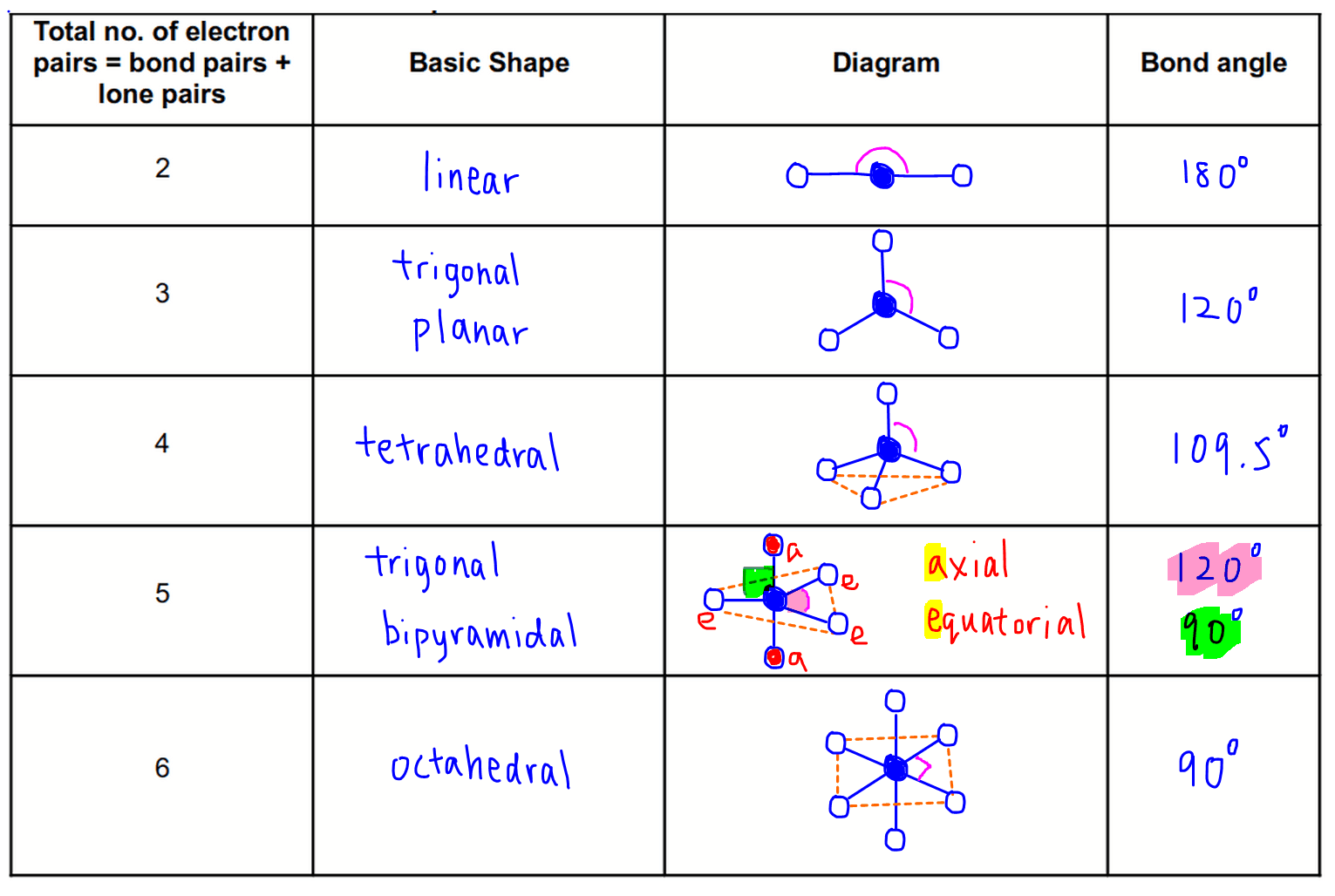
In SBr 2 there are 4 groups so it is tetrahedral. In SBr 2 this is 4, 2 S-Br bonds and 2 lone pairs = 4.Įlectron geometry is the geometry of all of the electron groups around the central atom. 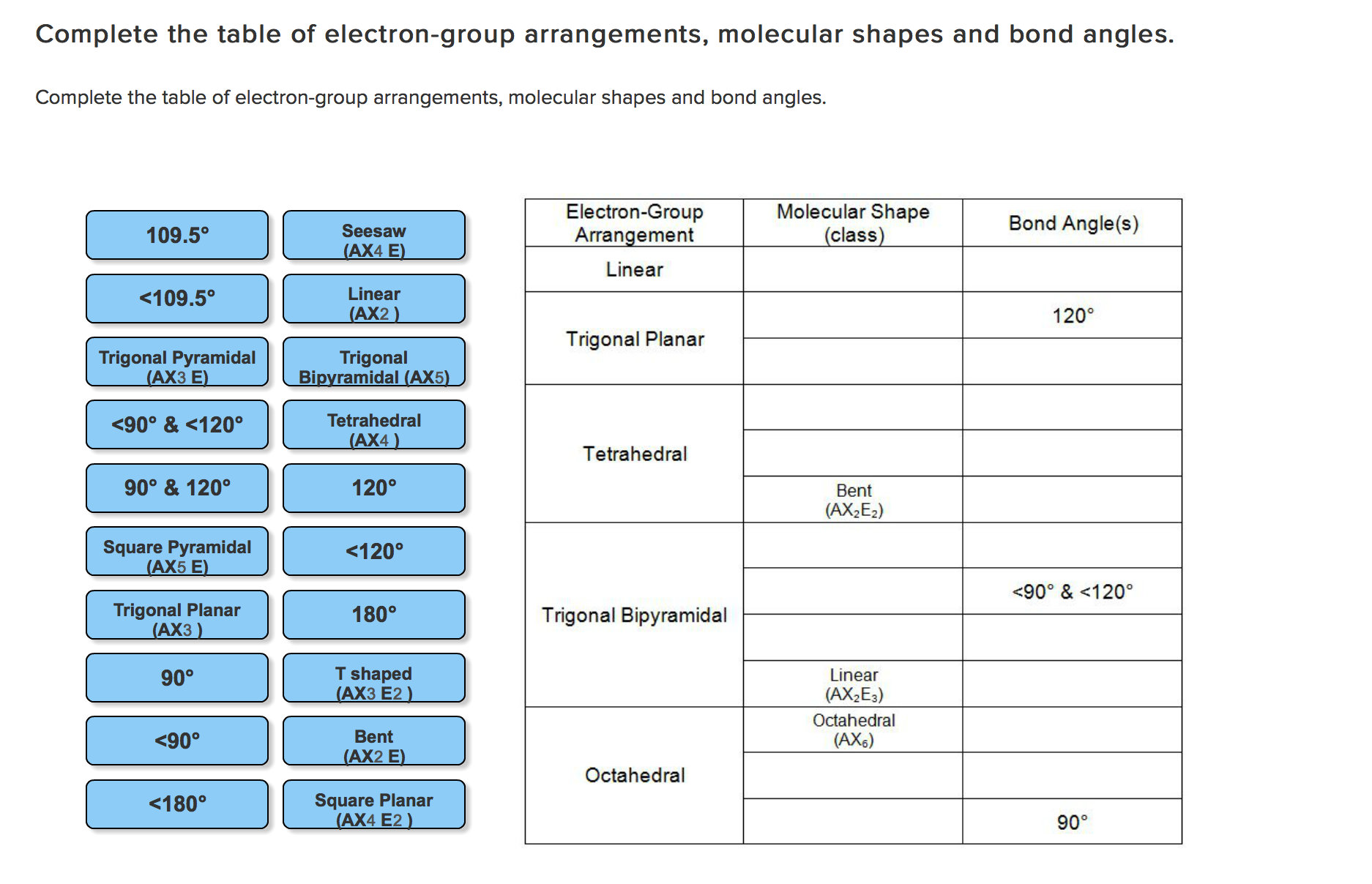
Total number of electron groups means the number of bonds and lone pairs around the central atom, but counting double and triple bonds as 1 "electron group". I think you've got it sorted but I figured I'd add this just in case. A screenshot is preferable to a picture of your laptop screen. Please do not ask for help acquiring, preparing, or handling illicit substances or for help with any activity that does not fall within the confines of whatever laws apply to your particular location.īonus points: If submitting a picture please make sure that it is clear. Any infractions will be met with a temporary ban at the first instance and a permanent ban if there is another. It is also important that you describe the specific part of the problem you are struggling with. It is OK if you are a little (or a lot!) stuck, we just want to see that you have made an effort. Please complete any questions as much as you can before posting. We will not do your homework for you, so don't ask. Please flair yourself and read over the rules below before posting. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
